Share This Article
Article Summary
U.S. Customs and Border Protection officers in Cincinnati intercepted more than 5,000 shipments of unapproved peptides linked to a coordinated smuggling operation. Senders primarily shipped the packages from China, mislabelingand concealing them within larger boxes to evade detection. Authorities identified over 300 related smuggling attempts beginning in December 2025. The FDA has not approved the seized substances, which may pose health risks to consumers.
CBP peptide seizure exposes large-scale smuggling operation at Cincinnati hub
The U.S. Customs and Border Protection (CBP) officers at the Port of Cincinnati reveal thousands of unapproved drug shipments entering the U.S.
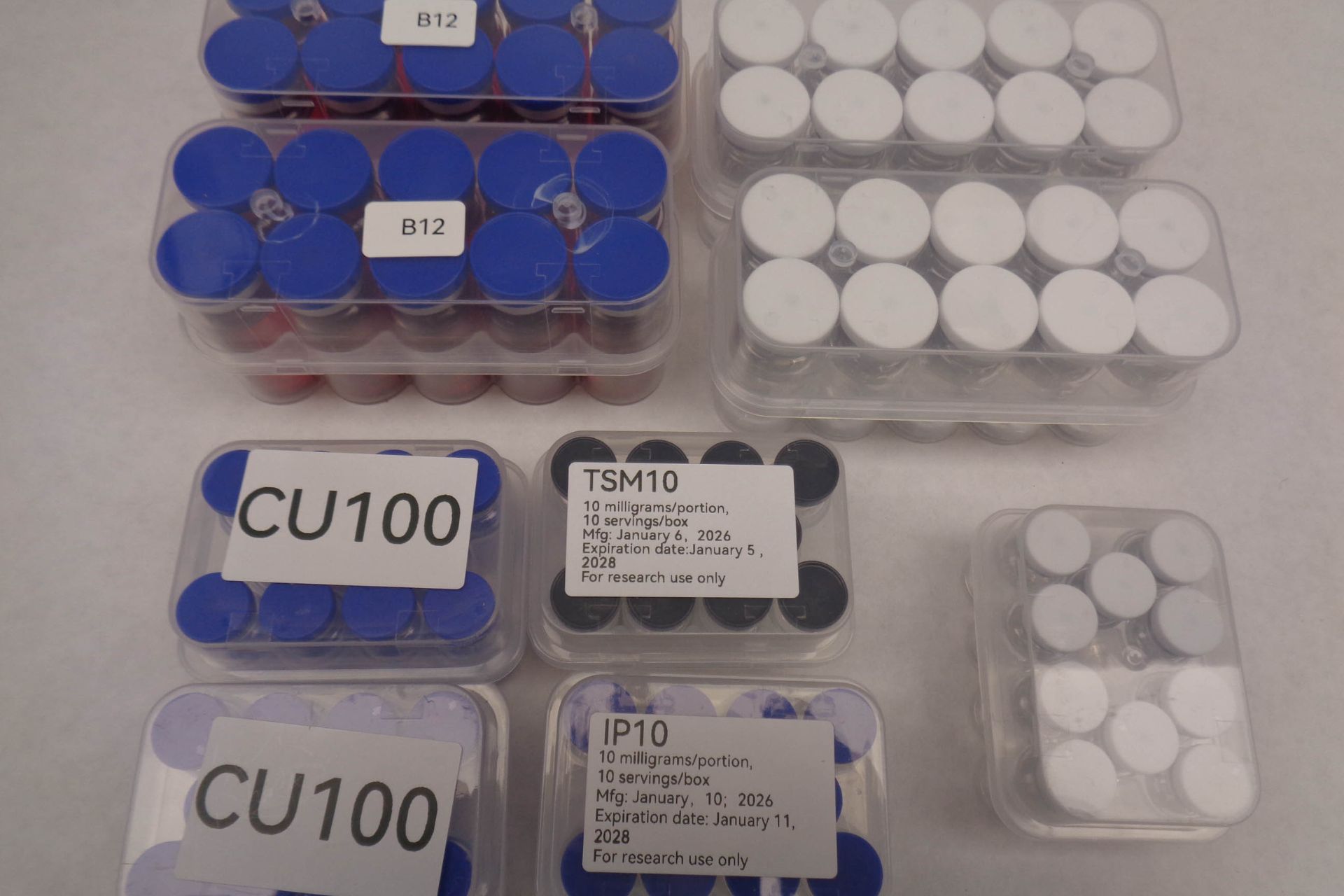
In a press release, the CBP said that the peptide seizure involved more than 5,000 packages containing unregulated peptides, many tied to popular weight-loss treatments.
They intercepted the shipments over several months, identifying what officials described as a complex and ongoing smuggling scheme.
According to the U.S. CBP, the operation began surfacing in December 2025 and quickly expanded in scope. Authorities reported more than 300 separate smuggling attempts connected to the same pattern of shipments.
The packages originated primarily in China and traveled through international mail systems to reach U.S. consumers. Officials said senders intentionally mislabeled the shipments to evade inspection and detection. They falsely declaredmany packages as harmless goods or low-value items.
According to reporting by WLWT and WHIO-TV, each large shipment often contained multiple smaller packages concealed inside, multiplying the total number of illegal items entering the system.
How the scheme worked
The said operation revealed a consistent smuggling method designed to bypass customs screening. Officers found that traffickers packed bulk boxes with layers of smaller shipments, each individually labeled for delivery.
Investigators identified several key tactics used in the scheme:
- Large outer boxes concealed 10–15 smaller packages
- False declarations masked the contents as benign goods
- Shipments were split into smaller quantities to reduce suspicion
- Multiple recipients across the U.S. were listed to distribute risk
This approach allowed smugglers to move significant quantities while attempting to avoid triggering enforcement thresholds. Accordingly, CBP officials noted that the structure of the operation suggested organized coordination rather than isolated incidents.
Furthermore, authorities emphasized that the Cincinnati port plays a major role in processing international e-commerce shipments, making it a frequent target for illicit imports. This is largely because the facility handles millions of packages annually, creating prime opportunities for bad actors to exploit both volume and speed.
What was inside the CBP peptide seizure shipments
Officials confirmed the seized products included peptides commonly associated with weight-loss and performance-enhancing drugs. Some of these compounds are linked to GLP-1 receptor agonists, a class of medications used in treatments similar to Ozempic and Wegovy.
However, the intercepted substances were not approved by the U.S. Food and Drug Administration. That means they were not legally permitted for import, sale, or distribution in the United States.
Health officials warn that unapproved peptides can pose serious risks. These include:
- Unknown or inconsistent dosage levels
- Contamination during manufacturing
- Lack of clinical testing or safety validation
- Mislabeling of active ingredients
CBP warns that consumers who purchase these products online may not realize they are receiving unregulated substances. Overseas sellers marketed many of the shipments to U.S. buyers seeking cheaper alternatives to prescription medications.
Public health concerns tied to peptide seizure
The seizure by CBP underscores growing concerns about the safety of imported pharmaceuticals and supplements. Officials say demand for weight-loss drugs has surged, creating opportunities for counterfeit and unapproved products to enter the market.
CBP warned that these substances can cause unpredictable side effects or fail to deliver intended results. In some cases, users may delay proper medical treatment by relying on unverified products.
Meanwhile, the Food and Drug Administration continues to advise consumers to purchase medications only through licensed pharmacies and approved providers. Imported drugs that bypass regulatory oversight may not meet U.S. safety standards.
Local coverage, including previous reporting by The Cincinnati Exchange on regional drug trends, has highlighted how illicit supply chains adapt quickly to consumer demand. In a related story, the outlet examined how underground markets respond to shortages and rising prices of prescription medications.
Ongoing investigation after CBP peptide seizure
While no charges have been officially filed in connection with the CBP peptide seizure, law enforcement confirms that the matter remains under active investigation. To facilitate this process, CBP stated it is working with federal partners to trace the origins and intended distribution networks connected to the shipments.
Specifically, officials are analyzing shipping data, recipient information, and payment methods to identify the individuals or organizations involved. The scale of the operation suggests a broader network that likely extends far beyond a single supplier.
In the meantime, the agency continues to monitor incoming shipments for similar patterns. Consequently, this increased scrutiny at ports of entry may lead to additional seizures in the coming months.
Ultimately, CBP encouraged the public to report suspicious packages or online sellers offering unapproved medical products. Enforcement efforts, officials noted, rely heavily on both proactive intelligence gathering and widespread public awareness.
Growing trend in illegal pharmaceutical imports
The Cincinnati case reflects a national trend involving illegal pharmaceutical imports through e-commerce channels. As online marketplaces expand, regulators face increasing challenges in policing cross-border shipments.
Experts say the combination of high demand, global supply chains, and digital storefronts creates a fertile environment for illicit trade. Small-package shipping, in particular, has become a preferred method for smuggling due to its volume and speed.
According to federal data cited in multiple reports, seizures of unapproved medical products have increased in recent years. The rise aligns with growing interest in weight-loss drugs and performance-enhancing compounds.
Here is a breakdown of recent, official statistics regarding the illegal importation of pharmaceuticals via e-commerce intercepted by U.S. Customs and Border Protection (CBP) in Cincinnati:
The Spring 2026 Peptide Bust (Dec 2025 – March 2026)
- Total Intercepted: Approximately 5,000 individual shipments of unapproved peptides from China.
- Smuggling Method: Over 300 “master cartons” were seized. Each large box contained roughly 15 smaller, pre-labeled packages designed to slip past customs as individual online orders.
+1 - Substances: The seizures heavily featured active ingredients for GLP-1 weight-loss drugs (semaglutide, tirzepatide, retatrutide) and strictly prohibited therapeutic peptides (MOTS-C, TB10, semax).
The Multi-Million Dollar Sweep (August 2025)
- Total Scope: Over a single, week-long joint operation with the FDA, Cincinnati CBP seized 54,843 unapproved or counterfeit medical products.
- Weight-Loss Specifics: Of the total items seized, 16,740 were specifically unapproved weight-loss medications, including counterfeit Ozempic and raw semaglutide/tirzepatide vials.
- Estimated Value: Had the items been legitimate, the combined value would have exceeded $3.53 million.
- Origins: The vast majority of these illicit shipments originated from Hong Kong, China, Colombia, and South Korea, destined for 40 different U.S. states.
Fall 2025 Targeted Operation (October 2025)
- Total Intercepted: Cincinnati officers seized 398 shipments containing 8,521 pairs of illicit contact lenses, alongside 50 shipments of misbranded drugs (GLP-1s, Botox, dermal fillers).
- Estimated Value: The cumulative Manufacturer’s Suggested Retail Price (MSRP) for the prohibited pharmaceuticals was $407,784.
Counterfeit Ozempic Bust (May 2024)
- Total Intercepted: Officers seized 11 targeted shipments containing over 100 injections each of counterfeit Ozempic.
- Estimated Value: The seizure was valued at approximately $887,000. The illicit medications were intercepted arriving from Colombia and were bound for addresses from New York to Texas.
FAQs
What did CBP seize in Cincinnati?
CBP officers seized more than 5,000 shipments of unapproved peptides entering the United States. Officials linked many of these substances to weight-loss and performance-enhancing drugs.
Where did the shipments originate?
Senders in China shipped most of the intercepted packages. They routed the items through international mail systems and addressed them to various recipients across the U.S.
Why are these peptides illegal?
The peptides were not approved by the FDA for use or distribution in the United States. Unapproved substances may lack safety testing, proper labeling, and quality control.
Is anyone facing charges in connection with the scheme?
Authorities have not announced any arrests related to the seizures. The investigation remains ongoing as officials work to identify those responsible.